
Summary
This paper examines why certain surgical robotic innovations succeed while others fail, using four analytical frameworks: Jobs-To-Be-Done Theory, Diffusion of Innovation, the Henderson-Clark Model, and Institutional Theory. Early systems like PUMA and ROBODOC demonstrated technical precision but failed commercially by solving unrecognized problems and lacking institutional legitimacy. AESOP succeeded by addressing universal pain points with minimal disruption, while da Vinci achieved market dominance through strategic beachhead targeting, comprehensive training infrastructure, and legitimacy across regulatory and professional domains. Telesurgery represents the next frontier but remains confined to demonstrations despite technical feasibility proven. Barriers include network latency, regulatory ambiguity around cross-jurisdiction practice, liability uncertainties, and absent institutional legitimacy. Strategic pathways exist through time-critical beachheads like acute stroke intervention, staged adoption from teleconsultation to full remote surgery, and building legitimacy through academic partnerships. Success requires simultaneous progress across technical, regulatory, economic, and institutional dimensions.
Presentation
The evolution of surgical robotics over the past four decades illustrates the complex relationship between technological possibility and the institutional realities of healthcare innovation. As Christensen et al. argue, disruptive innovations in medicine rarely diffuse on technical merit alone but succeed when they align with the “jobs” that stakeholders need to accomplish and when organizational structures are prepared to absorb their impact [1]. Early robotic platforms such as PUMA and
ROBODOC demonstrated remarkable precision yet struggled to achieve widespread adoption, revealing the limits of technological capability without user-centered value. By contrast, AESOP and the da Vinci Surgical System achieved commercial and institutional success. Examining the past, present, and emerging future of surgical robotics through theoretical lenses provides critical insight into how innovation in this domain advances and why certain systems reshape surgical practice while others remain historical curiosities.
Analysis Framework
As innovations succeed in different ways, it is necessary to establish frameworks for comparison. This paper employs four frameworks: Jobs-To-Be-Done (JTBD) by Clayton Christensen, Diffusion of Innovation Theory by Everett Rogers, the Henderson-Clark Innovation Model, and Institutional Theory.
The JTBD framework [2] posits that customers do not simply purchase products or services, but rather “hire” solutions to accomplish specific jobs in particular circumstances, encompassing functional, emotional, and social dimensions. Additionally, these “jobs” are context-dependent rather than universal. In the context of healthcare, a surgical robot might be hired by surgeons for ergonomic comfort, by hospitals for competitive differentiation, and by patients for faster recovery. This difference requires innovators to understand which jobs are most compelling for adoption decisions.
Rogers’ Diffusion of Innovation Theory [3] examines how innovations spread through social systems. Five attributes determine adoption rate: (1) relative advantage, (2) compatibility with existing practices, (3) complexity, (4) trialability, and (5) observability. Critically, perceived attributes matter more than objective performance, explaining why marginal technical advantages may diffuse rapidly if highly compatible and observable, while superior technologies may languish if perceived as complex.

The Henderson-Clark model [4] categorizes innovation by whether core components and architectural linkages change, creating four types: incremental, modular, architectural, and radical innovation. The key insight is that architectural innovations particularly challenge incumbent organizations because they disrupt embedded knowledge and established routines while appearing deceptively similar to existing technologies.
Institutional Theory posits that organizations adopt innovations for legitimacy within their institutional environment. DiMaggio and Powell [5] identified three isomorphic mechanisms: coercive (regulatory pressure), mimetic (peer imitation), and normative (professional standards). This framework is particularly relevant to healthcare, where regulatory approval, professional society endorsements, and peer adoption patterns powerfully shape technology diffusion independent of clinical superiority.
Early Development of Surgical Robots

On April 11, 1985, researchers integrated a Unimation PUMA 200 robot to perform the world’s first robotic surgery: a CT-guided stereotactic brain biopsy [6]. While stereotactic neurosurgery existed since 1905, manual head frame adjustment procedure was tedious, time-consuming, and might violate sterility requirement. These challenges motivated this robotics application that promised higher accuracy and versatility. Similarly, in the late 1980s, a PUMA 560 robot was used to perform prostatectomy [7].

Despite the success of these experimental cases, it was not until the early-to-mid 1990s that the FDA approved the first surgical robots for clinical use. ROBODOC, an autonomous milling system for hip and knee replacement, first operated in the United States in 1992 but received FDA commercial approval only in 2008 after performing 24,000+ procedures worldwide [8]. However, the majority of joint replacements remain manual [9]. AESOP (Automated Endoscopic System for Optical
Positioning), a robotic endoscope holder controlled by hand, foot, or voice, received FDA clearance in 1994 and achieved far greater adoption: 30,000 procedures in 300+ hospitals by 1998, exceeding 125,000 cases by the early 2000s [10, 11].

JTBD analysis reveals fundamental differences between these cases. PUMA addressed stereotactic biopsies with submillimeter precision: a narrow, infrequent functional job limiting market opportunity. ROBODOC addressed hip implant preparation with precision, but surgeons did not perceive existing manual techniques as inadequate, essentially solving an unrecognized problem. AESOP, however, addressed a universal, frequently experienced job: controlling the laparoscopic camera. Every surgeon performing laparoscopy encountered frustration with fatigued or inattentive camera holders, making this a high-frequency, high-pain problem addressing functional, emotional, and social dimensions of a job.
Diffusion of Innovation explains adoption spread differences. PUMA scored poorly across all attributes: minimal relative advantage, low compatibility, high complexity, no trialability, and limited observability. ROBODOC exhibited critical disconnect between objective and perceived attributes: measurable geometric precision remained largely invisible because it did not translate to observably superior clinical outcomes in most cases. Poor compatibility (requiring CT scans, preoperative planning, additional surgical time) and trialability (high capital costs demanding full commitment) created substantial friction. AESOP, on the other hand, excelled on every attribute: immediate visible advantage (steady camera versus trembling hands), seamless compatibility (no major workflow change), intuitive simplicity, high trialability, and obvious observability.
Henderson-Clark model fills in the gap for JTBD and Diffusion Theory, and reveals why certain innovations disrupt embedded knowledge. ROBODOC represented radical innovation, simultaneously changing components (autonomous milling replaced manual instruments) and architecture (CT-based workflows, altered OR configurations, redesigned team roles). This dual disruption destroyed tacit organizational knowledge underlying orthopedic expertise: haptic feedback, established division of labor, and procedural routines. Even surgeons recognizing ROBODOC’s job found themselves incompetent novices when their architectural knowledge became obsolete. AESOP, instead, represented modular innovation: it replaced only the camera holder while preserving the entire laparoscopic architecture. Surgeons maintained familiar positions, techniques, sequences, and communication patterns. This preservation explains why AESOP achieved high compatibility and easy adoption, by requiring minimal unlearning.
Institutional Theory fills the critical gap explaining ROBODOC’s marginalization despite proven technical merit. AESOP achieved legitimacy rapidly through all three isomorphic mechanisms: coercive (1994 FDA clearance), mimetic (300-hospital bandwagon once prestigious centers adopted), and normative (professional society endorsement, incorporation into training). ROBODOC suffered legitimacy crisis across all dimensions: delayed FDA approval until 2008 meant sixteen years of regulatory
limbo, only six hospitals by 1997 prevented mimetic isomorphism, and it never achieved normative legitimacy as orthopedic societies did not endorse it and residency programs excluded it. ROBODOC failed not only because it solved an unrecognized job (JTBD), scored poorly on diffusion attributes (Rogers), and disrupted organizational knowledge (Henderson-Clark), but fundamentally because it never achieved social legitimacy required for healthcare innovations to be professionally acceptable.
The Current State of Surgical Robotics Innovation
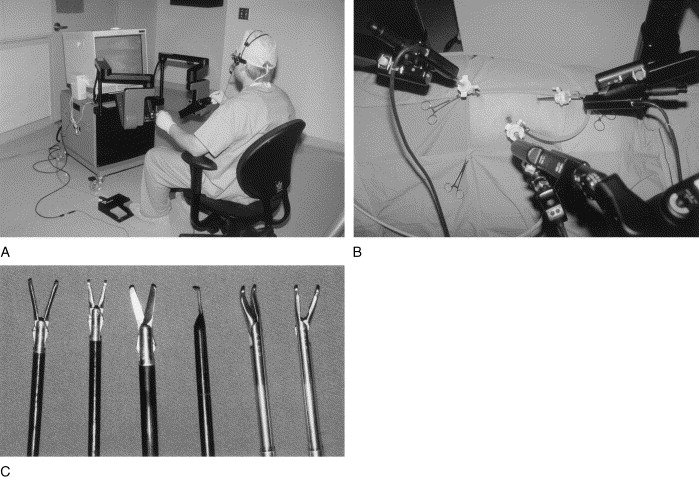
AESOP’s success demonstrated that surgical robotics could deliver tangible mainstream value beyond being experimental technology in academic research, catalyzing next-generation systems. Computer Motion (maker of AESOP) developed ZEUS, which added instrument manipulation arms to AESOP’s camera control, achieving FDA clearance in 2001 and international attention through Operation Lindbergh, the first transatlantic telesurgery in 2001 [12]. Meanwhile, Intuitive Surgical developed the da Vinci system with superior capabilities (3D visualization, articulating “wristed” instruments, enhanced ergonomics), receiving FDA approval in 2000 [13]. Competitive pressure between the two companies led to the 2003 merger, after which ZEUS was discontinued. Today, da Vinci dominates with 4,400+ global systems, representing near-monopoly status that has fundamentally reshaped minimally invasive surgery [14].
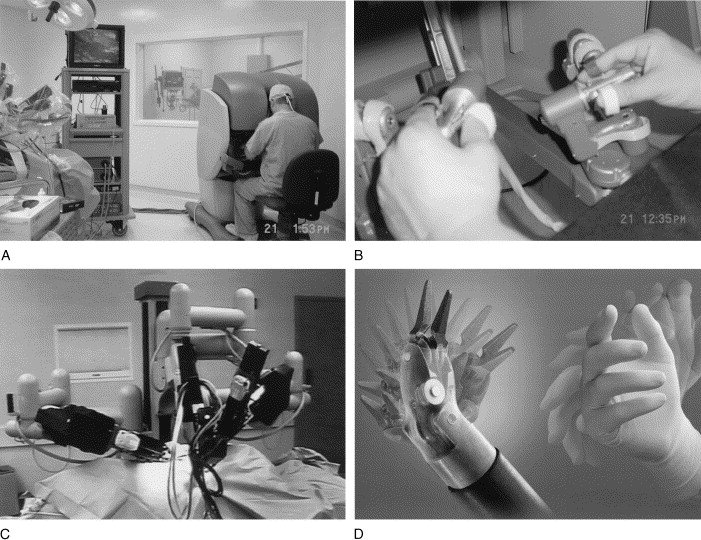
Da Vinci followed a “bowling pin” strategy, targeting prostatectomy as its beachhead after FDA approval in May 2001 [15]. Similar to how a bowling ball would first knock down the front pin (or “beachhead”), before the ball and that pin would knock down the remaining pins, this strategy starts with establishing dominance in specific surgical specialties before systematically expanding into adjacent markets [16]. Robotic-Assisted Laparoscopic Prostatectomy (RALP) cases exploded from 800 in 2003 to 8,000+ by 2004 [17], then expanded to gynecology and general surgery [18]. The razor-blade model ($1-2.5M systems plus recurring instrument/service costs) generated revenue funding continued R&D [19]. By 2017, 4,409 systems facilitated 877,000 procedures generating $3.1B revenue: 52% from instruments, 29% from systems, 19% from service [20]. The da Vinci name became synonymous with robotic surgery, with hospitals prominently marketing capabilities and patients specifically requesting “the robot.”
Taking a closer look, the system’s success can be attributed to its understanding of multiple, interconnected jobs that needed to be done across different stakeholder groups. For surgeons, it addresses functional (wristed instruments enabling confined-space surgery beyond conventional laparoscopy capability, 3D visualization, motion scaling reducing fatigue), emotional (professional prestige, burnout reduction), and social (career differentiation, competitive advantage) components [21]. For hospitals and administrators, competitive differentiation and marketing assets are connected to anxiety about being “left behind” once peers acquired systems [19]. For patients, it delivers functional benefits (faster recovery, less pain) plus emotional comfort of “most advanced” treatment and social cachet. The prostatectomy beachhead, moreover, proved strategic by targeting educated, affluent men deeply concerned about continence and sexual function preservation [22].
To understand how Intuitive reliably delivers these jobs at scale, one can look at their organization structure and product design. The company’s business model centered on a ’razor-and-blade’ revenue structure, with capital equipment sales generating only 29% of revenue while instruments and accessories (52%) and service contracts (19%) provided recurring income [20]. This model inherently creates strong incentives to maximize system utilization rather than simply selling capital equipment, on top of revenue generation. It’s logical to observe that the business model is centered on maximizing procedure volume as a north star metric, recognizing that successful job completion depends not just on system capability but on surgeon proficiency and institutional integration. Additionally, Intuitive tracked procedures per system as a key performance indicator, with the company reporting 877,000 procedures across 4,409 systems in 2017 [20], or an average of 199 procedures per system annually. To drive this utilization, Intuitive designed proprietary instruments with ten-use limitations, creating a direct financial link between procedure volume (a metric rooted in JTBD) and company success. The company also invested heavily in surgeon training infrastructure, establishing dedicated training centers and mandatory standardized credentialing pathways for surgeons. Intuitive’s training emphasized not just technical competence but psychological confidence, addressing the emotional job of reducing surgeon anxiety about complications during the learning phase. The system’s design embedded telemetry that transmitted anonymized surgical data back to Intuitive, allowing the company to analyze procedure patterns, identify best practices, and refine training curricula. This practice essentially creates a learning system that continuously improves job delivery. Marketing efforts focused on patient education, with Intuitive funding awareness campaigns, patient education websites, and hospital marketing materials that framed robotic surgery as a patient right rather than a physician preference, thereby ensuring the patient-driven demand job remained active. The company’s customer success teams worked directly with hospitals to optimize scheduling, improve OR efficiency, and maximize case volume, recognizing that the functional job of performing surgery was inseparable from the organizational context enabling high utilization. This comprehensive approach to job delivery explains why da Vinci succeeded: Intuitive understood that delivering the job required building an entire ecosystem, not just a capable robot.
The ingeniousness of Intuitive’s strategy can be further understood by looking at what the robot competes against. For specific procedures like prostatectomy and hysterectomy, da Vinci made complex minimally invasive surgery accessible to more surgeons. It competes simultaneously against open surgery (the prevalent approach, offering minimally invasive benefits as an advantage) and conventional laparoscopy (which few surgeons could perform for these procedures due to technical difficulty with straight instruments in confined spaces). This represented competition against constrained consumption: the procedures were happening, but technical limitations meant most surgeons defaulted to open approaches while only highly skilled laparoscopists could perform them minimally invasively. Da Vinci removed these technical barriers, essentially democratizing advanced minimally invasive surgery. In general surgery applications like cholecystectomy, however, da Vinci struggled precisely because it competed against highly optimized laparoscopy procedures that already solved the job well at much lower cost. The difference between the success of the two types of procedure, specific and general, reveals that the system only truly succeeded where existing solutions left significant jobs unaddressed [19].
From a Diffusion of Innovation perspective, da Vinci exhibited a more complex profile than its predecessors, scoring high on some attributes while initially struggling with others, yet strategically overcoming these limitations through focused market development. The system’s relative advantage was highly procedure-dependent, demonstrating clear superiority for prostatectomy and complex pelvic surgery while showing no benefit over conventional laparoscopy [19]. Da Vinci initially scored
low on compatibility, requiring dedicated operating room space, new team coordination patterns, and substantially longer setup times. The system’s complexity was significant, with learning curves estimated at 20-250 cases depending on procedure complexity [23]. However, da Vinci compensated for these weaknesses through exceptional observability: procedural footage could be recorded, shared at conferences, and used in marketing materials, making the technology’s capabilities highly visible to both professional and general audiences. The company’s training centers and credentialing pathways facilitated trialability despite high capital costs. The prostatectomy beachhead created a context where relative advantage was undeniable and procedure volume sufficient to justify investment, allowing early adopters to demonstrate success that later adopters could observe and emulate.
Henderson-Clark model reveals da Vinci succeeded as architectural innovation by managing knowledge disruption strategically. Like ROBODOC, da Vinci changed surgery’s architecture: surgeon-patient spatial relationship, hand-eye coordination, team communication, and workflows. However, it preserved more architectural elements: maintaining fundamental laparoscopic logic (small incisions, endoscopic visualization) that surgeons understood [21], while wristed instruments and 3D visualization restored open surgery elements (wrist articulation, depth perception) that conventional laparoscopy had removed. Targeting procedures where disrupted knowledge was less entrenched (laparoscopy relatively young versus hip replacement), establishing training programs, and providing compensating advantages also made disruption acceptable.
Institutional Theory provides the most crucial explanation for da Vinci’s dominance: comprehensive legitimacy through all three mechanisms creating self-reinforcing network effects. Coercive isomorphism operated through rapid FDA approvals (2000, 2001) [13, 15] without ROBODOC’s delay, with robotic prostatectomy incorporated into clinical guidelines creating normative pressure. Mimetic isomorphism proved explosive: once prestigious centers (Johns Hopkins, Memorial Sloan Kettering) adopted and published outcomes, community hospitals rushed to acquire systems, creating 4,400+ installations by 2017. Surgeons without training risked marginalization and referral losses [18]. Normative isomorphism cemented da Vinci’s position as professional societies endorsed robotic approaches, residency programs incorporated training, and board certification began expecting competency [23]. These three mechanisms created self-reinforcing cycles: regulatory approval enabled mimetic copying, widespread adoption created normative standards, and professional standards generated coercive pressure. The platform became institutionalized as the de facto standard, transforming “robotic surgery” into a near-synonym for da Vinci in professional discourse, patient expectations, and organizational routines.
Da Vinci’s dominance represents both culmination of decades of development and foundation for the next frontier. Successful surgical robotics require technical capability plus strategic alignment with user needs (JTBD), favorable diffusion characteristics (Rogers), manageable organizational disruption (Henderson-Clark), and comprehensive institutional legitimacy (Institutional Theory). Da Vinci achieved all four simultaneously, creating near-monopoly status [24, 14]. Notably, this dominance is predicated on a fundamental constraint: surgeon and patient must occupy the same physical space. Telesurgery challenges this by decoupling surgical expertise from physical presence entirely. This change represents an architectural leap far more radical than da Vinci’s console control, raising profound questions about whether framework patterns explaining past successes can predict remote surgical innovation’s trajectory.
Remote Robotics in Healthcare: Possibilities & Challenges
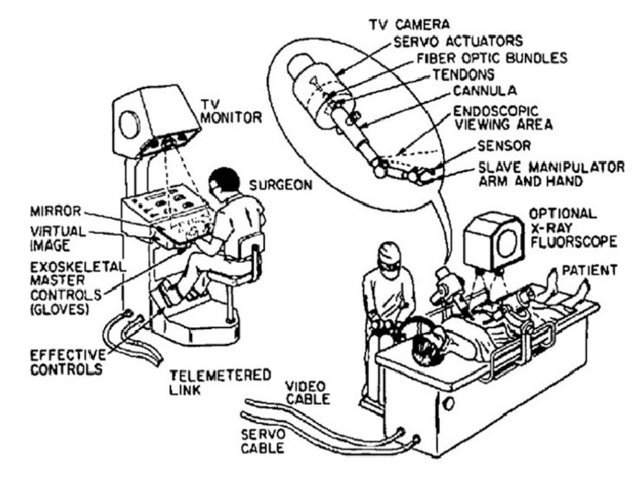
Remote surgery represents a natural evolution emerging from surgical robotics maturity and persistent geographic disparities in specialist access. Conceptual foundations trace to the 1970s, when NASA investigated surgical care for astronauts during long-duration space missions, recognizing that vast distances would require decoupling surgical expertise from physical presence [25]. To the present, as da Vinci demonstrated console-mediated control feasibility, the logical extension became apparent: if console/robot separation by several feet works, why not by thousands of miles? This notion gained urgency as rural healthcare faced critical specialist shortages: non-consumption where procedures could not happen due to geographic constraints rather than technical impossibility. The most dramatic validation of technical feasibility came on September 7, 2001, when Operation Lindbergh made history as the first transatlantic telesurgery. In this operation, professor Jacques Marescaux’s team in New York performed a laparoscopic cholecystectomy on a patient in Strasbourg, France, overcoming 7,000 kilometers with 155ms latency via dedicated fiber-optic infrastructure [12]. Between 2003 and 2007, Dr. Mehran Anvari demonstrated telesurgery could transition from demonstration to sustained practice, establishing the world’s first telerobotic service performing 20+ remote procedures between Hamilton and North Bay, Ontario, spanning 400 kilometers [26]. Additional military demonstrations reinforced potential for addressing non-consumption in underserved populations [27, 28]. These cases proved unequivocally that telesurgery could be done.
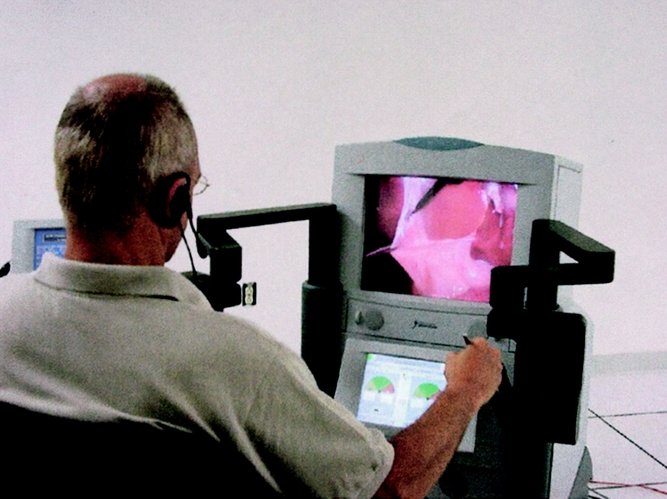
Telesurgery’s promise extends beyond technological novelty, addressing fundamental jobs across healthcare stakeholders that current solutions critically underserve. For rural hospitals, telesurgery provides specialist access without full-time employment, transforming economically unviable coverage into achievable capability. Patients in underserved regions currently face emotional and financial burdens traveling hundreds of miles for specialty care, often delaying time-critical treatment with serious health consequences. Telesurgery addresses receiving timely, expert care close to home. For specialist surgeons concentrated in urban centers, the technology extends expertise geographically without the travel time and lifestyle costs, potentially multiplying impact across multiple sites daily. Applications in military and disaster medicine can provide surgical capability where transporting patients is dangerous or impossible. Telesurgery could enable surgeons to operate while physically protected from infectious patients, a need highlighted during the COVID-19 pandemic. From a public health perspective, telesurgery addresses massive inefficiency of highly trained specialists geographically concentrated in urban centers while vast populations lack access. Unlike da Vinci’s competition against “good enough” alternatives in established markets, telesurgery competes primarily against non-consumption, in situations where necessary procedures simply do not happen due to geographic barriers to specialist access. This competitive positioning theoretically represents the most favorable condition for disruptive innovation, creating new market space without directly threatening established practices [2, 26, 29]. Yet if telesurgery addresses vast unmet needs and faces minimal competitive resistance, why has adoption remained stalled at isolated demonstrations rather than accelerating toward widespread deployment?
Despite technical feasibility and compelling jobs to be done, telesurgery faces formidable barriers that the four analytical frameworks reveal as fundamentally different from those encountered by previous surgical robotics innovations. From a technical perspective, network latency remains the most studied constraint: research demonstrates that surgical performance degrades significantly when latency exceeds 300-500 milliseconds, yet intercontinental distances via conventional internet infrastructure routinely produce delays exceeding this threshold [30]. While dedicated fiber-optic networks can reduce latency to acceptable levels, as Operation Lindbergh demonstrated, such infrastructure is prohibitively expensive for clinical deployment at scale. The absence of haptic feedback in current systems compounds latency issues: surgeons cannot feel tissue resistance, forcing complete reliance on visual cues and creating safety concerns during remote operation, where rapid physical intervention is impossible [31]. Network reliability poses another technical challenge: a momentary connection loss during remote surgery could prove catastrophic, yet achieving the “five nines” reliability (99.999% up-time) required for mission-critical medical applications demands significant infrastructure investments. Applying the Henderson-Clark framework, telesurgery represents an even more radical architectural innovation than da Vinci: it disrupts not only surgical technique but the fundamental spatial architecture of surgery itself, eliminating the surgeon’s physical presence and ability to intervene manually in emergencies. This architectural rupture threatens to obsolete organizational knowledge embedded in OR team coordination, crisis management protocols, and institutional muscle memory built around the surgeon’s bedside presence, creating knowledge disruption.
Regulatory and legal barriers create perhaps the most intractable obstacles, revealing through Institutional Theory why telesurgery lacks the coercive and normative legitimacy that enabled da Vinci’s success. In the United States, medical licensure operates on a state-by-state basis, creating ambiguity about whether surgeons performing remote operations across state lines require license in the patient’s state, the surgeon’s state, or both. This regulatory void translates to hospitals and surgeons unwilling to assume legal liability [32]. International telesurgery faces even greater complexity: no clear framework exists for cross-border medical practice, liability insurance, credentialing standards, or regulatory oversight when surgeon and patient occupy different legal jurisdictions. Insurance reimbursement policies remain unclear for telesurgical procedures, with payers questioning whether to reimburse based on the surgeon’s location, patient’s location, or even whether to cover remote procedures at all given unclear evidence of clinical benefit over local alternatives. The liability landscape is particularly murky: if complications arise during remote surgery, who bears responsibility? Is the onus on the remote surgeon, the local surgical team, the hospital, the robotic system manufacturer, or the telecommunications provider? These questions remain unanswered, creating a legitimacy vacuum that contrasts starkly with da Vinci’s rapid achievement of regulatory clarity and professional endorsement [33]. From an Institutional Theory perspective, telesurgery suffers from absent coercive isomorphism (no regulatory framework compelling adoption), absent mimetic isomorphism (too few successful examples for peers to copy), and absent normative isomorphism (professional societies have not endorsed it, training programs do not teach it, and it remains outside standard-of-care guidelines). Without institutional legitimacy across all three mechanisms, telesurgery cannot progress beyond isolated demonstrations regardless of technical capability.
Economic and adoption barriers compound these challenges, revealing through Rogers’ Diffusion framework why telesurgery scores poorly on attributes critical for adoption. The technology exhibits low compatibility with existing healthcare infrastructure: implementing telesurgery requires not just robotic systems but also dedicated high-bandwidth networks, redundant connectivity, specialized technical support, and coordination between geographically separated facilities [34]. Trialability is essentially non-existent. Unlike da Vinci, where hospitals could selectively use robotics for appropriate cases while maintaining conventional approaches for others, telesurgery demands full infrastructure investment before a single trial procedure. The complexity is extreme: beyond surgical technique, teams must master network management, cross-site coordination, emergency protocols for connection failure, and entirely new workflows. Observable benefits remain limited: while AESOP’s steady camera and da Vinci’s wristed instruments provided immediately visible advantages, telesurgery’s primary benefit (geographic access) is meaningful only for patients who would otherwise lack access, making the relative advantage invisible to most hospitals and surgeons who already have local expertise. From a JTBD perspective, the stakeholders who most acutely experience the job (rural patients and hospitals) lack the resources to invest in the solution, while stakeholders with resources (urban academic medical centers and specialist surgeons) experience the job less intensely, creating a misalignment between problem intensity and solution capacity.
With all these headwinds, identifying and executing a strategic entry point requires careful analysis of how innovative companies might navigate the technical, regulatory, economic, and institutional obstacles that have confined telesurgery to demonstration status for over two decades.
Towards Adoption of Telesurgery
While telesurgery faces tremendous barriers across dimensions, the framework analysis suggests that strategic pathways could enable progression from isolated demonstrations to clinical adoption. The key insight from da Vinci’s success is that breakthrough innovations do not require solving all problems simultaneously. One can succeed by identifying specific contexts, where value dramatically exceeds barriers, where early adoption becomes inevitable, and then leveraging that beachhead to expand systematically. For telesurgery, this means abandoning the notion of universal remote surgery as an entry point in favor of targeted applications where the jobs to be done are specific, and existing alternatives are inadequate, so that stakeholders will tolerate the technology’s current limitations and institutional ambiguity. The strategic question is not whether telesurgery can eventually replace all surgery, but rather which specific surgical scenarios create favorable conditions analogous to prostatectomy for da Vinci.
The most promising beachhead applications share three critical characteristics aligning with both JTBD framework and Rogers’ diffusion attributes: time-sensitivity making geographic access a life-or-death matter, high-value outcomes justifying infrastructure investment, and procedure characteristics compatible with current telesurgery capabilities. Acute stroke intervention exemplifies these criteria compellingly. Ischemic stroke treatment through mechanical thrombectomy must occur within hours of symptom onset to prevent permanent brain damage, yet approximately 50% of the US population lacks timely access to certified 24/7 mechanical thrombectomy-capable centers [35]. This focus presents a textbook case of non-consumption, where patients lose their lives or suffer permanent disability not from lack of medical knowledge but from geographic distance to expertise [28]. Telesurgery addresses a job that no alternative adequately solves: delivering specialist neurointerventional expertise to stroke patients in rural or community hospitals within the critical treatment window. The value proposition is stark: remote thrombectomy versus no thrombectomy, making relative advantage unambiguous in ways general surgical applications cannot match. Indeed, Lithuanian medical robotics company Sentante, founded in 2017, exemplifies this strategic focus. The company developed a haptic endovascular robotic platform specifically designed for remote thrombectomy procedures [36]. In October 2024, Sentante demonstrated capability through the world’s first transatlantic remote thrombectomy, where specialists in Jacksonville, Florida and Dundee, Scotland performed full end-to-end procedures on cadaveric models at the University of Dundee with only 120 milliseconds latency [37]. The FDA’s grant of Breakthrough Device Designation for Sentante’s stroke system in September 2024, with the company targeting 2026 market launch for peripheral interventions followed by stroke thrombectomy, suggests regulatory bodies recognize acute unmet need this application addresses [37, 38]. Similarly, trauma surgery in military or disaster contexts presents another high-value beachhead: when patients cannot be safely transported and local surgical capacity is overwhelmed or absent, telesurgery competes against death rather than conventional surgery, fundamentally altering risk-benefit calculation [39]. Rural emergency surgery for conditions like ruptured ectopic pregnancy or acute appendicitis requiring immediate intervention represents a third candidate, where telesurgery could enable community hospitals to retain emergency surgical capability rather than closing emergency departments due to inability to recruit surgeons, addressing hospital administrators’ existential job of maintaining facility viability [26].


These beachhead applications share an additional strategic advantage: they create conditions where regulatory and institutional barriers, while still formidable, become more negotiable. When the alternative to telesurgery is patient death or permanent disability, regulators and professional societies face different calculus than when telesurgery competes against functioning local alternatives. The FDA’s Breakthrough Device Designation for Sentante’s stroke system demonstrates this principle: when addressing life-threatening conditions with limited treatment options, regulatory pathways can accelerate [37]. States have demonstrated ability to authorize temporary or emergency licensure for out-of-state clinicians during capacity shortages, as widespread telehealth licensure waivers during COVID-19 created operational precedents, informing longer-term mechanisms like the Interstate Medical Licensure Compact [40, 41, 42]. In emergencies where the counterfactual is no treatment, liability expectations shift: crisis-standards-of-care frameworks and federal emergency protections, like the PREP Act, acknowledge altered standards and provide partial immunity for good-faith care [43, 44]. Most critically, successful outcomes in high-stakes cases generate observability necessary for mimetic isomorphism: dramatic saves of stroke patients through remote thrombectomy, documented in medical literature and covered by media, create compelling examples peer institutions can justify copying. Unlike ROBODOC’s invisible precision improvements, telesurgery successes in time-critical scenarios produce highly observable outcomes resonating with hospital administrators, surgeons, and the public: patients who walked out of hospitals who would otherwise have passed away or remained permanently disabled. Sentante’s transatlantic demonstration, involving reputable institutions and renowned specialists and generating significant media coverage, exemplifies how high-profile successes build awareness and legitimacy [36].
However, proceeding directly to fully remote telesurgery, even in optimal beachhead applications, likely underestimates architectural disruption and learning curves involved. Henderson-Clark suggests successful architectural innovations often stage transitions, allowing organizations to develop new knowledge systems incrementally rather than demanding immediate re-conceptualization. For telesurgery, this implies a gradual pathway building organizational capabilities, institutional legitimacy, and stakeholder confidence progressively. Initially, teleconsultation requires minimal infrastructure (standard videoconferencing) and creates no regulatory ambiguity, as the remote specialist provides advice while a local surgeon retains full control and legal responsibility. This addresses real jobs (access to specialist expertise for complex case planning) while requiring no new institutional frameworks, allowing hospitals and surgeons to develop comfort with remote collaboration. Then, telementoring and teleproctoring introduce robotic systems but maintain the local surgeon as primary operator while the remote specialist observes, guides, and can temporarily assume control during critical moments through dual-console capability [29]. This configuration preserves the local surgeon’s physical presence for emergency intervention, dramatically reducing safety concerns while allowing gradual skill transfer and confidence building. Telementoring has already achieved some adoption where high-volume centers support lower-volume sites, establishing proof points informing regulatory frameworks and insurance policies [27]. Only after organizations develop fluency with remote collaboration and regulatory precedents emerge from telementoring should full telesurgery, where the remote surgeon operates independently without local surgical backup, be attempted.
The economic and institutional barriers, while perhaps most intractable, are not insurmountable if approached strategically. High infrastructure costs make most sense when applied in high-volume applications (frequent use spreads costs over many operations) or when addressing high-value time-critical outcomes (value of rapid intervention outweighs infrastructure expenses), rather than routine elective surgery. A hub-and-spoke model, where a single tertiary care center provides telesurgical support to multiple rural hospitals, creates economies of scale making per-procedure costs more comparable to patient transport alternatives. As Sentante CEO Edvardas Satkauskas articulated: “This is about access and consistency. Access, because patients in remote or non-specialist hospitals can receive the gold-standard treatment without delay” [37]. Rural hospitals might justify telesurgery investment not through procedure volume but through strategic value beyond individual procedure reimbursement (JTBD for hospital administrators): ability to retain emergency surgical capability prevents ED closure, maintains hospital viability, and preserves community healthcare access [26]. Insurance reimbursement ambiguity could be addressed by initially targeting self-pay or government-funded demonstration projects establishing outcomes data and precedents, rather than waiting for comprehensive reimbursement policies. ARPA-H’s recent initiatives seeking proposals for automated neurovascular systems to broaden stroke treatment access signal governmental recognition and potential funding mechanisms for validation studies [ 38]. Medicare demonstration projects specifically focused on rural healthcare access could also serve as entry points, generating data and institutional experience necessary for broader policy development.
Building institutional legitimacy requires explicit recognition that telesurgery currently lacks the coercive, mimetic, and normative isomorphism that enabled da Vinci’s success, and that these cannot be achieved simultaneously but must be built sequentially. The initial goal should be establishing coercive legitimacy through regulatory clarity, even if limited: Sentante’s FDA Breakthrough Device Designation represents exactly this type of foundational regulatory validation, creating defensible
ground for clinical adoption [37]. Such designations signal regulatory bodies view the technology as addressing unmet needs, providing hospitals and surgeons confidence that approval pathways exist. Once a handful of institutions implement successful telesurgery programs under regulatory blessing, mimetic isomorphism can begin: peer hospitals can justify adoption by referencing established precedents rather than pioneering into regulatory ambiguity. Academic medical centers, which face less financial pressure and have missions encompassing innovation, represent ideal early adopters whose participation confers legitimacy community hospitals can later invoke. Sentante’s collaboration with prestigious institutions and involvement of renowned specialists exemplifies this strategy of building legitimacy through association with respected medical centers and practitioners [36]. Publishing outcomes data in peer-reviewed surgical journals, presenting results at professional society meetings, and engaging specialty organizations such as American College of Surgeons, Society for Vascular Surgery, and Society of NeuroInterventional Surgery in developing best practice guidelines can begin building the sense that telesurgery represents legitimate surgical practice [23]. Professional societies’ endorsement follows rather than precedes adoption; the strategic task is creating enough successful examples that societies feel compelled to provide guidance rather than avoidance.
Ultimately, unlocking telesurgery requires accepting that the pathway to adoption will likely be longer, more constrained, and more incremental than entrepreneurs or advocates might prefer. No single strategy overcomes all previously mentioned barriers. Thus, success likely requires simultaneous progress on multiple fronts: identifying and proving value in narrow beachhead applications, staging technological introduction from teleconsultation through telementoring to full telesurgery, developing technical solutions that work within current infrastructure constraints, building regulatory precedents through demonstration projects and breakthrough designations, and cultivating institutional legitimacy through academic partnerships and professional society engagement. The twenty-year gap between Operation Lindbergh’s demonstration and continued lack of widespread adoption suggests that technical feasibility, while necessary, represents only a small fraction of what successful innovation requires. However, companies like Sentante, with their strategic focus on acute stroke intervention as a beachhead, staged rollout beginning with peripheral interventions, technical innovations addressing key barriers like haptic feedback, and early regulatory validation through FDA Breakthrough Device Designation, demonstrate that thoughtful navigation of the multidimensional challenge space is possible. The frontier of remote robotics will advance not through technological breakthroughs alone, but through strategic clarity about where, how, and in what sequence to introduce capabilities that fundamentally challenge how surgery has been practiced throughout human history.
Final Words
Taken together, the historical progression from early experimental robots to contemporary surgical platforms reveals that successful innovation in robotic surgery depends on far more than technical advances. Systems like AESOP and the da Vinci platform prevailed because they aligned with well-defined stakeholder jobs, achieved favorable diffusion attributes, minimized architectural disruption, and accumulated institutional legitimacy across regulatory, professional, and organizational domains. These same analytical frameworks help explain why telesurgery, despite compelling demonstrations such as Operation Lindbergh and emerging remote endovascular platforms, has not yet transitioned into routine clinical practice. Its adoption is constrained by technical requirements, regulatory and licensure ambiguity, unresolved liability structures, and the absence of the coercive, mimetic, and normative forces that previously propelled robotic systems into the mainstream. Yet the trajectory outlined in this paper suggests that telesurgery may follow a similar phased pathway: beginning with teleconsultation, progressing through telementoring, and ultimately achieving full remote operation in high-acuity, high-value applications where the alternative is non-consumption. As disparities in surgical access persist and remote-capable robotic platforms mature, the future of innovation in surgical robotics will increasingly depend on the strategic alignment of technical capability, stakeholder need, and institutional readiness, just as it has throughout its past.
References
- C. M. Christensen, R. Bohmer, and J. Kenagy, “Will disruptive innovations cure health care?” Harvard Business Review, vol. 78, no. 5, pp. 102–112, 2000.
- C. M. Christensen, T. Hall, K. Dillon, and D. S. Duncan, Competing against luck: The story of innovation and customer choice. Harper Business, 2016.
- E. M. Rogers, Diffusion of innovations, 5th ed. New York: Free Press, 2003.
- R. M. Henderson and K. B. Clark, “Architectural innovation: The reconfiguration of existing product technologies and the failure of established firms,” Administrative Science Quarterly, vol. 35, no. 1, pp. 9–30, 1990.
- P. J. DiMaggio and W. W. Powell, “The iron cage revisited: Institutional isomorphism and collective rationality in organizational fields,” American Sociological Review, vol. 48, no. 2, pp. 147–160, 1983. [Online]. Available: http://www.jstor.org/stable/2095101
- Y. Kwoh, J. Hou, E. Jonckheere, and S. Hayati, “A robot with improved absolute positioning accuracy for ct guided stereotactic brain surgery,” IEEE transactions on biomedical engineering, vol. 35, no. 2, pp. 153–160, 1988.
- B. L. Davies, R. D. Hibberd, W. S. Ng, A. G. Timoney, and J. E. Wickham, “The development of a surgeon robot for prostatectomies,” Proceedings of the Institution of Mechanical Engineers, Part H: Journal of Engineering in Medicine, vol. 205, no. 1, pp. 35–38, 1991, describes preliminary feasibility study using PUMA 560 robot for transurethral resection of the prostate.
- “Digimatch robodoc surgical system receives u.s. fda 510(k) marketing clearance,” Aug 2008. [Online]. Available: https://www.meddeviceonline.com/doc/digimatch-robodoc-surgical-system-receives-0001
- R. Nichols, “The legacy of robodoc,” Sep 2022. [Online]. Available: https://www.comstocksmag.com/qa/legacy-robodoc
- “Clinical testing of aesop 3000; surgical robot assists minimally invasive surgery,” Industrial Robot: the international journal of robotics research and application, vol. 25, no. 3, 06 1998. [Online]. Available: https://doi.org/10.1108/ir.1998.04925cab.009
- J. Damiano, Ralph J. and H. S. Maniar, “An overview of the computer motion system,” Operative Techniques in Thoracic and Cardiovascular Surgery, vol. 6, no. 3, 08 2001.
- J. Marescaux, J. Leroy, F. Rubino, M. Smith, M. Vix, M. Simone, and D. Mutter, “Transcontinental robot-assisted remote telesurgery: Feasibility and potential applications,” Annals of surgery, vol. 235, no. 4, pp. 487–492, 2002.
- Food and Drug Administration, “FDA Approves New Robotic Surgery Device,” ScienceDaily, July 2000, fDA clearance for da Vinci Surgical System for general laparoscopic surgery. [Online]. Available: https://www.sciencedaily.com/releases/2000/07/000717072719.htm
- K. Shah and R. Abaza, “Diffusion and adoption of the surgical robot in urology,” Translational Andrology and Urology, vol. 9, no. Suppl 2, pp. S120–S125, March 2020, states “As new robotic systems enter the market, Intuitive’s monopoly on robotic surgery may dwindle”.
- Intuitive Surgical Inc., “Form 10-K Annual Report,” U.S. Securities and Exchange Commission, 2001, fDA clearance for laparoscopic radical prostatectomy received in May 2001. [Online]. Available: https://isrg.intuitive.com/static-files/9404a44d-3af9-44ed-9da5-44f5fa0b7b15
- G. A. Moore, Crossing the chasm : marketing and selling high-tech products to mainstream customers, rev. ed. ed. New York: HarperBusiness, 1999.
- L. R. Schachter, M. R. Kaufman, and S. D. Herrell, “Chapter 10 – establishment of a robotic prostatectomy program,” in Robotics in Urologic Surgery, J. A. SMITH and A. TEWARI, Eds. Philadelphia: W.B. Saunders, 2008, pp. 79–84. [Online]. Available: https://www.sciencedirect.com/science/article/pii/B9781416024651500146
- J. E. Anderson, D. C. Chang, J. K. Parsons, and M. A. Talamini, “The first national examination of outcomes and trends in robotic surgery in the united states,” Journal of the American College of Surgeons, vol. 215, no. 1, pp. 107–114, 2012.
- G. I. Barbash and S. A. Glied, “New technology and health care costs: The case of robot-assisted surgery,” New England Journal of Medicine, vol. 363, no. 8, pp. 701–704, 2010.
- C. P. Childers and M. Maggard-Gibbons, “Estimation of the acquisition and operating costs for robotic surgery,” JAMA, vol. 320, no. 8, pp. 835–836, 2018.
- A. R. Lanfranco, A. E. Castellanos, J. P. Desai, and W. C. Meyers, “Robotic surgery: A current perspective,” Annals of Surgery, vol. 239, no. 1, pp. 14–21, 2004.
- D. R. Yates, C. Vaessen, and M. Rouprêt, “From leonardo to da vinci: The history of robot-assisted surgery in urology,” BJU International, vol. 108, no. 11, pp. 1708–1714, 2011.
- D. M. Herron, M. Marohn, and S.-M. R. S. C. Group, “A consensus document on robotic surgery,” Surgical Endoscopy, vol. 22, no. 2, pp. 313–325, 2008.
- A. Lin, “Intuitive surgical, a company that essentially monopolizes surgical robots,” Andy Lin’s Long-term Stock Investment Blog, September 2024, reports Intuitive Surgical having about 80% of the surgical robotics market. [Online]. Available: https://www.granitefirm.com/blog/us/2023/08/17/intuitive-surgical-2/
- R. M. Satava, “Surgical robotics: The early chronicles – a personal historical perspective,” Surgical Laparoscopy, Endoscopy & Percutaneous Techniques, vol. 12, no. 1, pp. 6–16, 2002.
- M. Anvari, C. McKinley, and H. Stein, “Establishment of the world’s first telerobotic remote surgical
service,” Annals of Surgery, vol. 241, no. 3, pp. 460–464, 2005. - B. Challacombe and P. Dasgupta, “Telementoring and telerobotic surgery,” Current Urology Reports, vol. 4, no. 2, pp. 114–119, 2003.
- P. J. Choi, R. J. Oskouian, and R. S. Tubbs, “Telesurgery: Past, present, and future,” Cureus, vol. 10, no. 5, p. e2716, 2018.
- N. Raison, M. S. Khan, and B. Challacombe, “Telementoring and robot-assisted surgery,” Journal of Robotic Surgery, vol. 9, no. 3, pp. 127–130, 2015.
- S. Xu, M. Perez, K. Yang, C. Perrenot, J. Felblinger, and J. Hubert, “Determination of the latency effects on surgical performance and the acceptable latency levels in telesurgery using the dv-trainer® simulator,” Surgical Endoscopy, vol. 28, no. 9, pp. 2569–2576, 2014.
- A. M. Okamura, “Haptic feedback in robot-assisted minimally invasive surgery,” Current Opinion in Urology, vol. 19, no. 1, pp. 102–107, 2009.
- G.-Z. Yang, J. Cambias, K. Cleary, E. Daimler, J. Drake, P. E. Dupont, N. Hata, P. Kazanzides, S. Martel, R. V. Patel et al., “Medical robotics—regulatory, ethical, and legal considerations for increasing levels of autonomy,” Science Robotics, vol. 2, no. 4, p. eaam8638, 2017.
- N. Naik, B. Hameed, D. K. Shetty, D. Swain, M. Shah, R. Paul, K. Aggarwal, S. Ibrahim, V. Patil, K. Smriti et al., “Legal and ethical consideration in artificial intelligence in healthcare: Who takes responsibility?” Frontiers in Surgery, vol. 9, p. 862322, 2022.
- R. Rayman, K. Croome, N. Galbraith, R. McClure, R. Morady, S. Peterson, S. Smith, A. Van Wynsberghe, S. Primak, R. Patel et al., “Long-distance robotic telesurgery: A feasibility study for care in remote environments,” International Journal of Medical Robotics and Computer Assisted Surgery, vol. 2, no. 3, pp. 216–224, 2006.
- A. P. Kansagra, M. S. Goyal, S. Hamilton, and G. R. Cutter, “Access to mechanical thrombectomy for ischemic stroke in the united states,” Stroke, vol. 52, no. 7, pp. 2554–2561, 2021, population-wide study showing fewer than 50% of ischemic stroke patients initially received care at facilities capable of mechanical thrombectomy.
- N. Walter, “Transatlantic robotic stroke surgery performed across 4,000 miles,” Interesting Engineering, October 2024, details Sentante’s haptic feedback system and 120ms latency achievement. [Online]. Available: https://interestingengineering.com/ai-robotics/remote-robotic-thrombectomy-sentante-breakthrough
- Sentante, “Critical stroke procedure performed from across the atlantic,” October 2024, announces first transatlantic remote thrombectomy and FDA
Breakthrough Device Designation. [Online]. Available: https://sentante.com/milestone-for-remote-surgery-as-critical-stroke-procedure-performed-from-across-the-atlantic/ - Fierce Biotech, “Arpa-h aims to fund the development of autonomous stroke-treating robots,” November 2024, reports on governmental initiatives for stroke treatment robotics. [Online]. Available: https://www.fiercebiotech.com/medtech/arpa-h-launches-program-fund-autonomous-stroke-treating-robots
- T. P. Cundy, N. E. Gattas, G.-Z. Yang, and A. Darzi, “Penetrating trauma and the role of telemedicine in austere environments,” World Journal of Surgery, vol. 38, no. 7, pp. 1560–1565, 2014.
- Federation of State Medical Boards, “States waiving telehealth licensure requirements in response to covid-19,” 2020, accessed via FSMB COVID-19 resources.
- Centers for Medicare & Medicaid Services, “Medicare telemedicine health care provider fact sheet,” March 2020, public Health Emergency telehealth policy expansion.
- Interstate Medical Licensure Compact Commission, “Interstate medical licensure compact: Overview and participation,” 2021, background on multistate licensure framework.
- Association of American Medical Colleges, “Faq: Crisis standards of care,” December 2020, guidance on altered standards and liability context in emergencies.
- U.S. Department of Health and Human Services, “Declaration under the public readiness and emergency preparedness (prep) act for medical countermeasures against covid–19,” 2020, federal emergency liability protections.
- Carrier, James, “The henderson-clark innovation model: A simple summary,” PeopleShift, July 2019. [Online]. Available: https://people-shift.com/articles/the-henderson-clark-innovation-model/
- Takács, D. Nagy, I. Rudas, and T. Haidegger, “Origins of surgical robotics: From space to the operating room,” vol. 13, pp. 13–30, 01 2016.
- E. I. AB, Frame for Leksell Stereotactic System. Science Museum Group. [Online]. Available: https://collection.sciencemuseumgroup.org.uk/objects/co503283/frame-for-leksell-stereotactic-system
- P. Aidan and M. Bechara, “Gasless trans-axillary robotic thyroidectomy: The introduction and principle,” Gland Surgery, vol. 6, pp. 229–235, 06 2017.
- G. T. Sung and I. S. Gill, “Robotic laparoscopic surgery: a comparison of the da vinci and zeus systems,” Urology (Ridgewood, N.J.), vol. 58, no. 6, pp. 893–898, 2001.
- Sentante, “Sentante stroke system receives fda breakthrough device designation!” September 2025. [Online]. Available: https://sentante.com/sentante-stroke-system-receives-fda-breakthrough-device-designation/
- ——, “First fully robotic remote endovascular stroke thrombectomy!” June 2025. [Online]. Available: https://sentante.com/first-fully-robotic-remote-endovascular-stroke-thrombectomy/
